The Comparison of Different Diatom Digestive Method using HCl-H2O2 and HCl -KMnO4 in Telaga Pengilon Dieng
DOI:
https://doi.org/10.51264/inajl.v1i1.5Keywords:
Diatom, digest, methods, HCl, KMnO4, H2O2Abstract
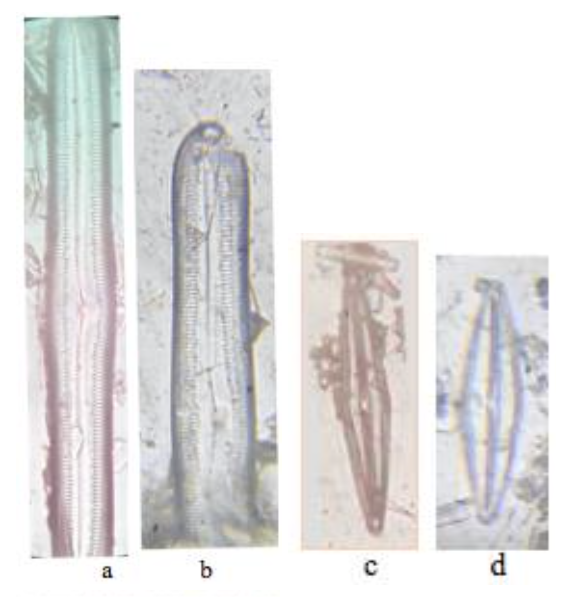
Identification diatom has been applied as an important key to tracing paleoenvironmental conditions. The knowledge for diatom extraction from sediment is continuing, but the diatom digestive methods in the tropical area is still limited. Telaga Pengilon, located in Dieng Indonesia, is rich in organic material in the sediment, the sample also associates with epiphytic and benthic diatom. Therefore, extracting pure diatom frustules free of organic matter from the sediments is essential for this application. To find the appropriate method for extracting diatoms in Telaga Pengilon, this research compares HCl with H2O2 and HCl with KMNO4 as the digestive reagent. Based on the statistical result (HCl, KMnO4) less time consumed than (HCl, H2O2) during the digestive process (p<0.05) and produced the same amount of residue (p> 0.05). For Pinnularia and Frustulia, the majority of diatoms could be extracted using both methods. Under microscopy, the structure of diatoms remained almost perfect after digestion with H202, the striae clearly visible and the residue digested correctly. Another method using HCl, KMnO4 fragmentation was found for some Pinnularia, and for Frustulia, the striae are clearly visible. This study demonstrated that different diatoms have different resistance for reagents and some of the locations need different reagents because different materials are contained in the sediment and need help to remove material organically. As far as the HCl and H2O2 are appropriate digestion methods in a tropical area such as Telaga Pengilon, HCl, KMnO4 can be a substitute for the less time-consuming process.
References
Antoniades D, Hamilton PB, Douglas MSV, Smol JP. 2008. Diatom of North America: The freshwater floras of Prince Petrcik, Ellef Ringnes, and Northern Ellesmere Island from Canadian Arctic Archipelago. Iconographia Diatomologica, 17.
Ajay R & Sakshi M. 2018. Significance of Diatoms in Diagnosis of Drowning Deaths: A Review, Journal of Forensic & Genetic Science.
Battarbee RW. 1973. A new method for estimating absolute microfossil numbers, with special reference to diatoms. Limnology and Oceanography, 18, 647–653.
Battarbee RW.1986. Diatom analysis. In: Berglund, B.E. (ed.) Handbook of Holocene Paleoecology and Paleohydrology, Wiley & Sons, London, 527–570.
De Tommasi E. 2016. Light manipulation by single cells: the case of diatoms. Journal of Spectroscopy.
Fucci N, Pascali V, Puccinelli C, Marcheggiani S, Mancini L, and Marchetti D. 2015. Evaluation of two methods for the use of diatoms in drowning cases. Forensic Science, Medicine, and Pathology, 11(4), 601-605.
Galbraith ED. 2006. Interactions between climate and the marine nitrogen cycle on glacial-interglacial timescales. Ph.D. Thesis, Univ. of British Columbia.
Griffiths K. 2015. High-latitude responses of diatom and chironomid assemblages to regional climate changes and other environmental stressors. Canada: Queen’s University Kingston, Ontario, Canada.
Hasle GR & Syvertsen EE. 1997. Marine diatoms, in Tomas CR (Ed.) Identifying Marine Phytoplankton, 5-385.
Hobbs WE, Wolfe P, Inskeep WP, Amskold A, and Konhuser KO. 2009. Epipelic diatoms from an extreme acid environment: Beowulf Spring, Yellowstone National Park, USA. Nova Hedwigia, 135, 71-83.
Krammer K & Lange-Bertalot H. 1991b. Su?sswasserflora von Mitteleuropa. Bacillariophyceae, Teil 3: Centrales, Fragilariaceae, Eunotiaceae. Jena. Gustav Fischer Verlag, p 576.
Krammer K & Lange-Bertalot H. 2004. Bacillariophyceae 4. Teil: Achnanthaceae, Kritische Erganzungen zu Navicula (Lineolatae), Gomphonema Gesamtliteraturverzeichnis Teil 1-4 [2nd revised edition] [With "Erga?nzungen und Revisionen" by H. Lange Bertalot]. In: H. Ettl et al., Suesswasserflora von Mitteleuropa. Spektrum Akademischer Verlad Heidelberg, 2(4), 1-468.
Krammer K & Lange-Bertalot H. 1991c. Bacillariophyceae. 3. Teil: Centrales, Fragilariaceae, Eunotiaceae. - In: ETtl H, Gerloff J, Heynig H, M Ollenhauer D. (Eds): Su?sswasserflora Von Mitteleropa Fischer Verlag, Stuttgart, vol. 2/3, pp 1-576. ISBN 978-3-8274-1987-3
Krammer K & Lange-Bertalot H. 1991d. Su?sswasserflora von Mitteleuropa. Bacillariophyceae, Teil 3: Centrales, Fragilariaceae, Eunotiaceae. Jena. Gustav Fischer Verlag, pp 1-576. ISBN: 978-93-83083-77-0
Krammer K & Lange-Bertalot H. 1991a. Bacillariophyceae. 4. Teil: Achnanthaceae, Kristische Erga?nzungen Zu Navicula (Lineolate) Und Gomphonema. In: ETtl H, Gerloff J, Heynig H, M Ollenhauer D. (Eds): Su?sswasserflora Von Mitteleropa Fischer Verlag, Stuttgart, vol 2/4, p 437.
Levin EA, Morgan RM, Scott KR, and Jones VJ. 2017. The transfer of diatoms from freshwater to footwear materials: An experimental study assessing transfer, persistence, and extraction methods for forensic reconstruction. Science & Justice.
Morales LV, Sigman DM, Horn MG, and Robinson RS. 2011. Cleaning methods for the isotopic determination of diatom-bound nitrogen in non-fossil diatom frustules. Limnology And Oceanography: Methods, 11,101-112.
Nagy SS. 2011 Collecting, Cleaning, Mounting, and Photographing Diatoms. In: Joesph Seckbach PK, editor. The Diatom World. Springer, New York, pp 5-11.
Renberg I. 1990. A procedure for preparing a large set of diatom slides from sediment cores. Journal of Paleolimnology, 4, 87-90.
Rohn EJ & Frade PD. 2006. The Role of Diatoms in Medicolegal Investigations I: The History, Contemporary Science, and Application of the Diatom Test for Drowning. Forensic Examiner, 15(3).
Round FE, Mann DG, and Crawford RM. 2007. Diatoms: Biology and Morphology of the Genera. Cambridge, p 747.
Ruhland K, Karst T, Paterson A, Eaves GR, Smol JP, and Cumming BF. 1999. Standard Sediment Sample Preparation Methods for Siliceous Microfossils (Diatoms and Chrysophyte Scales and Cysts). Paleoecological Environmental Assessment and Research Laboratory (PEARL), Department of Biology Queen’s University.
Schowe KA. 2012. Diatom communities across a gradient of acid mine drainage on the West Coast, South Island, New Zealand. Thesis. Master of Science in Environmental Science, University of Canterbury.
Serieyssol K, Chatelard S, and Cubizolle H. 2010. Diatom fossils in mires: a protocol for extraction, preparation, and analysis in palaeoenvironmental studies. International Mire Conservation Group and International Peat Society, 12, 1-11.
Soeprobowati TR, Hadisusanto S, and Gell P. 2012. The diatom stratigraphy of Rawapening Lake, Implying Eutrophication History. Am J Env Sci 2012, 8(3), 334-344.
Soeprobowati TR, Suedy SWA, and Hadiyanto. 2016. Diatom and water quality of Telaga Warna Dieng, Java, Indonesia. IOP Proceeding. Earth and Environmental Sciences, 55, 1-6.
Soeprobowati TR & Suedy SWA. 2018. Find the future from the past: Paleolimnology in Indonesia. E3S Web of Conferences, 31, 08002. EDP Sciences.
Taylor JC & Cocquyt C. 2016. Diatom from the Congo and Zambesi Basins. Methodologies and identification of the Genera. ISSN 1784-1291.
Taylor JC, Harding WR, Archibald CGM. 2007. An Illustrated Guide to Some Common Diatom species from South Africa. Water Research Commission, pp 178. ISBN 1-77005-484-7.